GyaniMed is a local-first AI operating system. It connects domain-specific agents to your lab tools: laboratory software, DoE and Stats software, HPLC, GC, Particle sizer, Microscope, Spectrophotometer, DSC and runs the analysis for you. Air-gapped (No internet needed) by design. No cloud dependency.
The bottleneck is not the experiment. it's everything around it.
Every instrument comes with specialist software that takes months to learn. Once trained, the work is still manual: export from the instrument, open the analysis tool, copy the outputs, paste into a report. Each step is a context switch and a chance for error. Senior scientists end up buried in operational overhead instead of doing science.

ELNs are static. Cloud AI has IP risk. RAG stops at information access. What is missing is an execution layer: software that actually does the work.
Excellent systems of record. But static; they store data after work is done. They do not operate software or run analyses.
Powerful general intelligence. But high risk: IP leakage concerns, data egress violations, and no control of local desktop tools in regulated labs.
Improved retrieval. But passive; it stops at information access. Offers no task completion, no execution, no structured output.
Agents that operate your equipment software directly. Run the analysis. Generate the report. Produce the audit trail. Without needing Internet. Deterministically.
Scientist types: “Generate a dose-response curve and calculate EC50 from the latest assay.” Natural language. No menus. No training.
Agent identifies the correct dataset. Selects the right tool (statistical analysis, DoE modelling, instrument controller). Configures analysis parameters. Operates the software automatically via Agentic connectors.
Agent generates the curve, extracts metrics, produces traceable output. Full GxP audit log generated automatically; every step recorded, reproducible, and ready for regulatory review.
Each agent understands the scientific domain it operates in: the data structures, the workflows, and the compliance requirements. They execute with statistical rigor, not LLM guesswork.
Physicochemical characterization, solubility profiling, stability screening, excipient compatibility. Determines whether a molecule can become a viable dosage form.
LNP optimization: encapsulation efficiency, particle size distribution, PDI, zeta potential. Batch-to-batch variability analysis with multivariate parameter comparison across iterative runs.
Full factorial, fractional factorial, and response surface designs. Automated ANOVA. Operates statistical analysis and DoE platforms directly, extracting data, computing analysis, and detecting statistically significant differences.
Optimization workflows for injectable formulations, oral solid dosage, and complex delivery systems. Links process parameters to quality attributes with full traceability.
Bridge bench-to-pilot across solvent systems, mixing speeds, shear forces, and equipment constraints. Identifies critical process parameters that shift at scale before they become GMP failures.
Evidence-bound deviation reporting. Traceable study comparison. GxP audit trail generation. Every agent action logged with timestamps and reproducible workflow records for regulatory review.
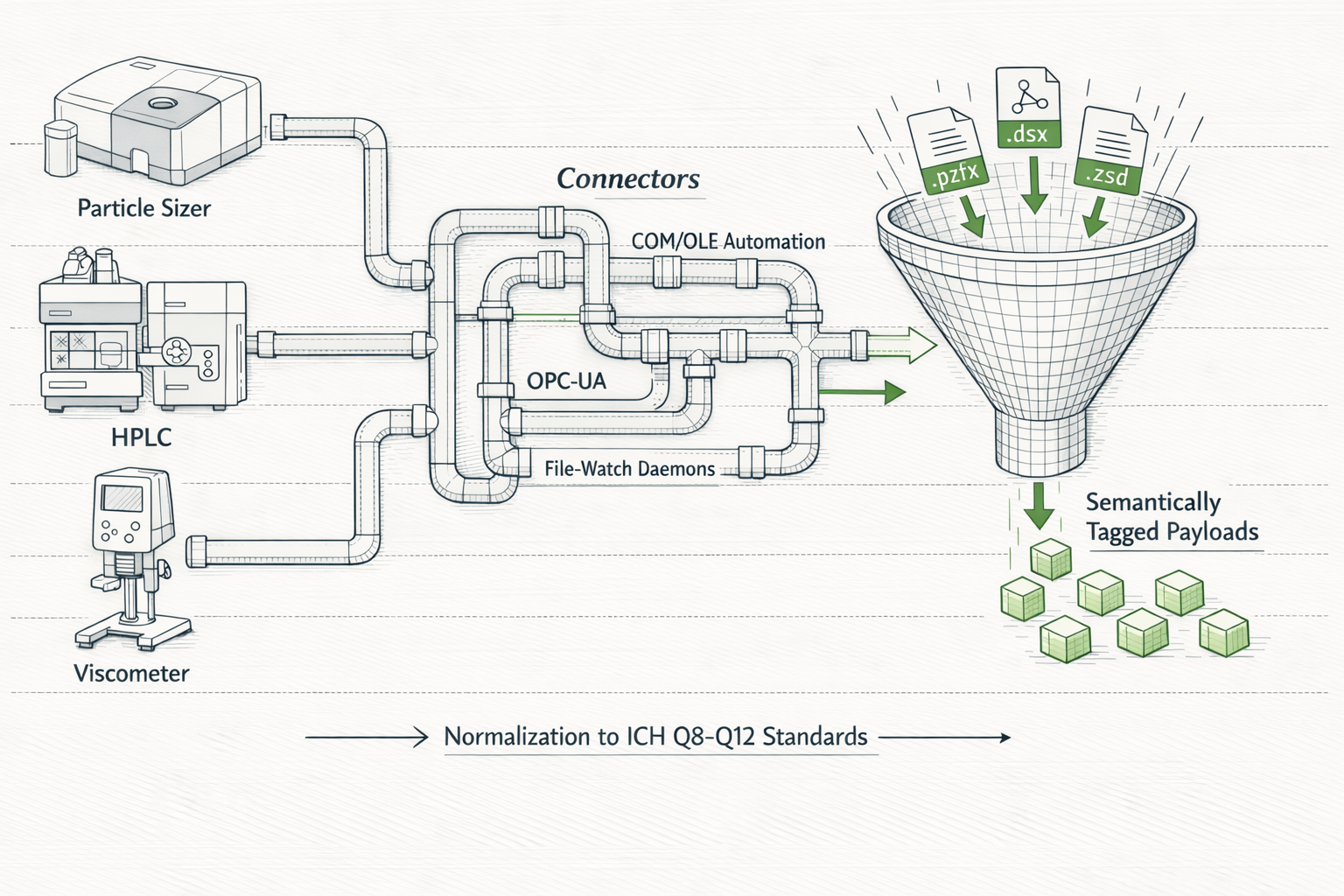
Agents operate lab tools through Connectors, the integration protocol that replaces copy-paste workflows.
Each connector enables an agent to directly control a specific software tool or instrument output. Add a new connector → increase platform utility → deepen institutional integration → increase switching costs. The connector ecosystem is the flywheel that makes GyaniMed irreplaceable.
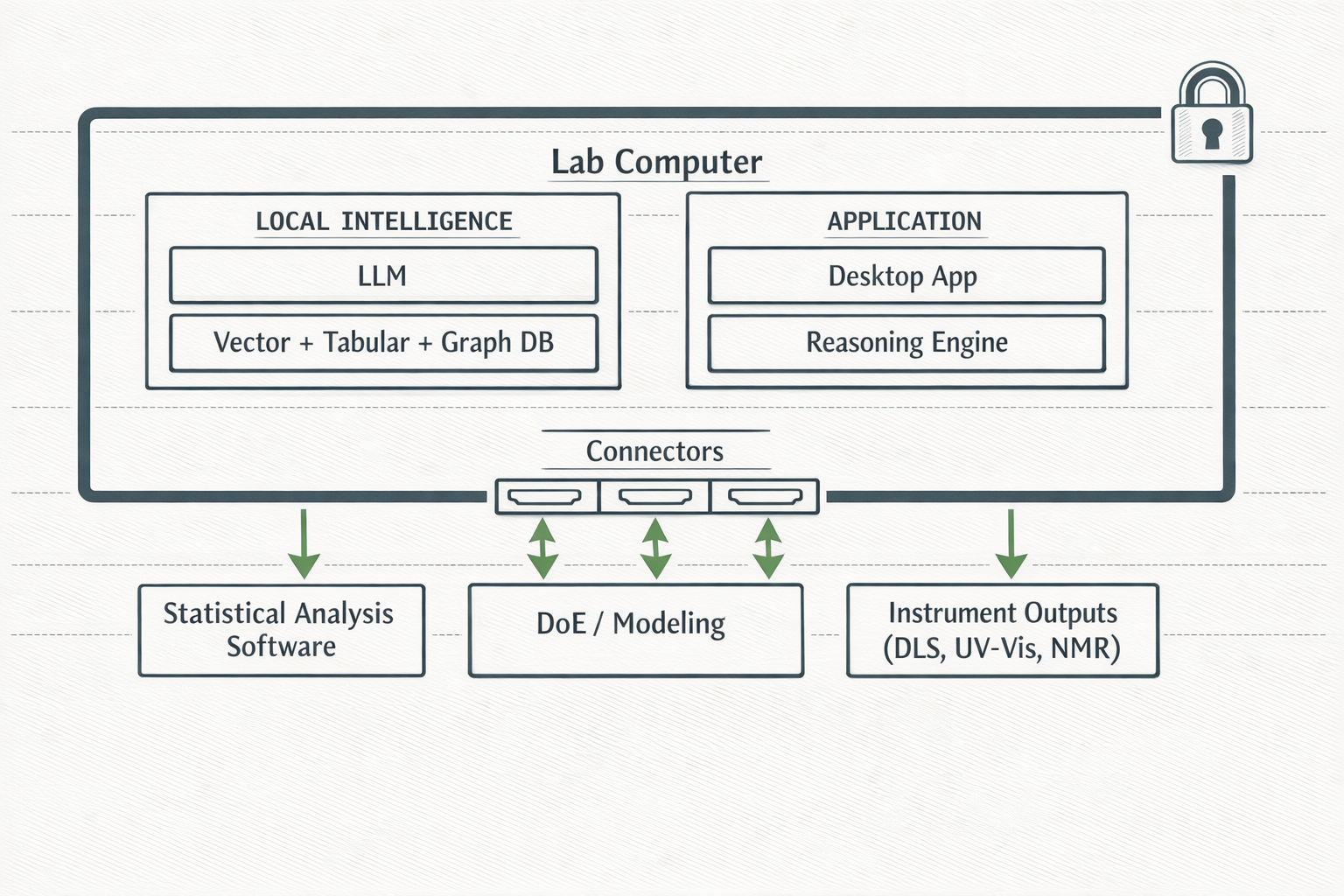
This is not an “on-prem option.” Local-first is the default architecture. Data processing happens entirely on-device. Retrofitting cloud architectures to work locally is technically prohibitive; GyaniMed is native to the edge.
Local LLM and ML models running on-device. Vector, tabular, and graph databases. Orchestration engine manages all agent routing, tool selection, and workflow execution without any cloud call.
Standardized protocol for controlling external tools. Each connector maps agent intent to tool-specific operations. Statistical analysis, DoE modelling, chromatography, and instrument outputs are all accessible through a single orchestration interface.
Graph + Vector + Tabular Database working in unison. Natural language queries simultaneously access all three sources. QbD relationships, experimental embeddings, and structured batch data are queried together.
Every agent action logged with timestamps. Every step recorded. Workflows can be re-run exactly. Full trails for regulatory review. In GxP-adjacent workflows, black-box AI is unacceptable; GyaniMed ensures complete traceability.
GyaniMed operates in the translational gap between molecule and clinical trial, where formulation engineering meets regulatory accountability. High complexity, high audit burden, fragmented tooling.
Many AI tools recommend. GyaniMed executes. That distinction is rare and valuable.
Retrofitting cloud architectures to work locally is technically prohibitive. GyaniMed is native to the edge, with an estimated 18–24 month replication timeline for competitors.
Each new AI Connector increases platform utility, deepens institutional integration, and increases switching costs. The ecosystem compounds with every tool connected.
2,400+ domain-specific mappings. CQAs, CMAs, CPPs, QbD relationships encoded as first-class entities. This is not generic metadata; it is pharmaceutical intelligence built into the schema.
Competitors are cloud-dependent. Regulated pharma blocks data egress. GyaniMed operates where cloud-native AI cannot go: inside the firewall, on the scientist’s own machine.
Agents. Connectors. Skills. The operating system for pharmaceutical R&D.
Analyses in seconds, not days of manual copy-paste across tools
Every action logged. Every result traceable. GxP-grade from day one.
All data stays on-device. Air-gapped. No cloud egress. No IP leakage.
Expert knowledge captured, structured, and reusable. No more tribal knowledge loss.
AI Agent Operating System for pharmaceutical research and manufacturing. Local-first. Air-gapped. Domain-specific agents. Connectors. Built for scientists, by scientists.
Get in touch →Zero-setup pilot. Instant visibility. Phase 1 FIH readiness.